Chlorthalidone: Thiazide-like Diuretic
Overview
Generic name (Active ingredient): Chlorthalidone
Drug class: Thiazide-like diuretic
Dosage forms and strengths: Oral tablets: 12.5 mg, 50 mg
Indications
Chlorthalidone is a long-acting thiazide-like diuretic primarily used for the management of hypertension and various forms of edema. Its prolonged duration of action—lasting up to 48–72 hours—makes it one of the most effective options among diuretics for blood pressure control.
1. Hypertension (High Blood Pressure): Chlorthalidone is widely prescribed as first-line therapy for primary hypertension, either alone or in combination with other antihypertensive agents such as ACE inhibitors, ARBs, beta-blockers, or calcium channel blockers. Clinical evidence suggests that chlorthalidone offers superior cardiovascular protection compared with hydrochlorothiazide, particularly in reducing risks of stroke and heart failure.
2. Chronic Heart Failure (NYHA Class II–III): Used as adjunct therapy in patients with mild to moderate chronic heart failure to alleviate fluid retention and reduce preload and afterload, thereby improving symptoms such as dyspnea and edema.
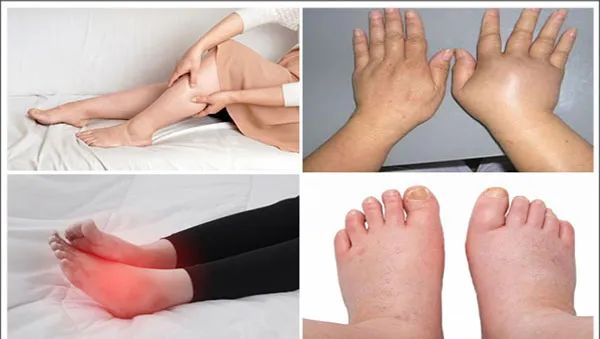
3. Edema of Various Origins:
Chlorthalidone helps manage fluid accumulation associated with:
- Congestive heart failure
- Cirrhosis of the liver
- Corticosteroid or estrogen therapy
- Nephrotic syndrome
- Acute glomerulonephritis
- Chronic renal insufficiency
4. Ascites due to Cirrhosis: For patients with compensated cirrhosis and ascites, chlorthalidone can be used under strict medical supervision to promote diuresis and control fluid buildup, provided that electrolyte balance is carefully monitored.
5. Diabetes Insipidus (Nephrogenic Type): Chlorthalidone can paradoxically reduce urine output in patients with diabetes insipidus by inducing mild volume depletion, which enhances water reabsorption in the proximal tubules.
6. Prevention of Calcium Kidney Stones: By reducing urinary calcium excretion, chlorthalidone lowers the risk of recurrent calcium oxalate or calcium phosphate nephrolithiasis, especially in patients with idiopathic hypercalciuria.
Pharmacodynamics
Chlorthalidone is a long-acting diuretic structurally related to benzothiadiazines (thiazides). Although not a true thiazide, it shares a nearly identical mechanism of action and therapeutic profile.
Chlorthalidone acts primarily on the distal convoluted tubule of the nephron, where it inhibits sodium and chloride reabsorption by blocking the Na⁺/Cl⁻ cotransporter. This inhibition leads to an increase in sodium, chloride, and water excretion. As a result of greater sodium delivery to the collecting ducts, there is enhanced potassium and hydrogen ion excretion, which can occasionally cause hypokalemia or metabolic alkalosis.
Interestingly, chlorthalidone also enhances calcium reabsorption in the distal tubules, which contributes to its usefulness in preventing calcium-containing kidney stones.
When used as monotherapy, chlorthalidone effectively lowers blood pressure in patients with mild to moderate hypertension. It is particularly effective in elderly individuals and has been shown to reduce the incidence of stroke, coronary artery disease, and overall cardiovascular morbidity and mortality.
In nephrogenic diabetes insipidus, chlorthalidone produces a paradoxical antidiuretic effect, decreasing polyuria. Although the exact mechanism is not fully understood, it is believed to result from mild extracellular fluid volume depletion, which increases proximal sodium and water reabsorption.
Pharmacokinetics
Absorption
Chlorthalidone is moderately absorbed from the gastrointestinal tract, with an oral bioavailability of approximately 64%. Peak plasma concentrations are typically reached 8–12 hours after ingestion, consistent with its slow onset and prolonged action.
Distribution
The drug is highly protein-bound (~76%), mainly to albumin, and exhibits extensive distribution into body tissues. It can cross the placental barrier and is excreted into breast milk, necessitating caution in pregnancy and lactation.
Metabolism
Chlorthalidone undergoes limited metabolism. It is eliminated primarily unchanged, both through the biliary route into feces and via renal excretion in urine. Approximately 70% of an administered dose is excreted within 120 hours, mostly in its unchanged form.
Elimination
Renal excretion is the major elimination pathway. Chlorthalidone has a prolonged elimination half-life of about 50 hours, which remains stable even after long-term use—accounting for its once-daily dosing schedule and sustained antihypertensive efficacy.
Toxicity and Overdose
Drug Interactions
Interactions with Other Medications
Neuromuscular blockers and antihypertensives: Chlorthalidone may enhance the effects of curare-derived muscle relaxants and other blood pressure-lowering medications, increasing the risk of hypotension.
Corticosteroids, ACTH, β₂-agonists, amphotericin B, and carbenoxolone: These agents can intensify hypokalemia induced by chlorthalidone, potentially leading to muscle weakness or cardiac arrhythmias.
Antidiabetic drugs: Thiazide-like diuretics may reduce the hypoglycemic effect of insulin and oral antidiabetic agents. Dosage adjustments may be necessary.
Digitalis (digoxin): Thiazide-induced hypokalemia or hypomagnesemia can increase the risk of digitalis-induced arrhythmias.
NSAIDs (nonsteroidal anti-inflammatory drugs): Concomitant use may reduce the diuretic and antihypertensive effects of chlorthalidone by inhibiting renal prostaglandin synthesis.
Anticholinergic agents (e.g., atropine, biperiden): May increase the bioavailability of thiazide diuretics by reducing gastrointestinal motility and delaying gastric emptying.
Anion-exchange resins (e.g., cholestyramine): Can decrease chlorthalidone absorption, reducing its therapeutic effect.
Allopurinol, amantadine, diazoxide, cytotoxic agents:
- May increase hypersensitivity reactions to allopurinol.
- Amantadine toxicity may be enhanced.
- Diazoxide-induced hyperglycemia may be potentiated.
- Renal clearance of cytotoxic drugs may be reduced, heightening their myelosuppressive effects.
Calcium salts and vitamin D: Concurrent use can increase serum calcium levels, occasionally leading to symptomatic hypercalcemia (manifesting as fatigue, muscle weakness, or anorexia), particularly in patients with hyperparathyroidism.
Cyclosporine: Co-administration may increase uric acid levels and the risk of gout.
Lithium: Thiazide-like diuretics reduce renal clearance of lithium, potentially causing toxic serum concentrations. Concurrent use is not recommended.
ACE inhibitors and potassium-sparing diuretics: Combining these with chlorthalidone can lead to severe hyperkalemia. Caution and close monitoring are advised.
Contraindications
Hypersensitivity to chlorthalidone, sulfonamide derivatives, or any component of the formulation
Anuria
Severe hepatic or renal impairment (creatinine clearance < 30 mL/min)
Persistent hypokalemia, hyponatremia, or symptomatic hypercalcemia
Gout or hyperuricemia
Pregnancy-related hypertension
Untreated Addison’s disease
Concurrent lithium therapy
Dosage and Administration
Adults
Hypertension:
- Initial: 25 mg once daily, preferably in the morning
- May increase to 50 mg/day if needed
- Maximum dose: 100 mg/day
Chronic, stable heart failure (NYHA class II–III):
- Start with 25–50 mg/day
- In severe cases, may increase up to 100–200 mg/day
- Maintenance: 25–50 mg daily or every other day
Edema of known etiology:
- Initial: 50–100 mg/day, or 100 mg every other day / 3 times weekly
- May increase to 150–200 mg/day if necessary
- Maintenance: Adjust to the lowest effective dose
- Maximum: 200 mg/day
Nephrogenic diabetes insipidus:
- Initial: 100 mg twice daily
- Maintenance: 50 mg/day, adjusted according to clinical response
Pediatric dosing:
- Use the lowest effective dose.
- Typical initial dose: 0.5–1 mg/kg every 48 hours
- Maximum: 1.7 mg/kg every 48 hours
Special populations
Elderly: Use lower starting doses and monitor closely for electrolyte disturbances.
Renal impairment: Ineffective when creatinine clearance < 30 mL/min. Avoid use.
Administration guidelines:
- Administer orally once daily, preferably with breakfast.
- Use the lowest effective individualized dose to minimize adverse effects.
Adverse Effects
Very common: Hypokalemia, hyperuricemia, hyperlipidemia (especially at high doses)
Common:
- Hyponatremia, hypokalemia, hyperglycemia
- Skin rash, urticaria
- Orthostatic hypotension
- Dizziness, lightheadedness
- Loss of appetite, mild dyspepsia
- Erectile dysfunction
Uncommon: Gout
Rare:
- Hypercalcemia, glycosuria, or worsening diabetes control
- Photosensitivity reactions
- Cholestatic jaundice or hepatic dysfunction
- Cardiac arrhythmias
- Headache, paresthesia
- Nausea, vomiting, abdominal pain, constipation, or diarrhea
- Thrombocytopenia, leukopenia, agranulocytosis, eosinophilia
- Idiopathic pulmonary edema, allergic reactions
- Interstitial nephritis
Very rare: Hypochloremic alkalosis
Frequency not known: Choroidal effusion
Precautions and Warnings
Use cautiously in patients with hepatic impairment or progressive liver disease—risk of hepatic encephalopathy, especially in cirrhotic patients.
In renal impairment, accumulation may occur with prolonged use; monitor urea and creatinine levels.
Sulfonamide derivatives, including chlorthalidone, may rarely cause choroidal effusion, acute myopia, or secondary angle-closure glaucoma. Symptoms (blurred vision, eye pain) may appear within hours to weeks after initiation. Untreated cases can result in permanent vision loss.
Risk factors include a history of allergy to sulfonamides or penicillin.
Monitor electrolytes regularly, particularly in the elderly, cirrhotic patients, or those with nephrotic syndrome.
In nephrotic syndrome, use only under strict medical supervision and in patients without hypovolemia.
When initiating ACE inhibitors, consider reducing or discontinuing chlorthalidone for 2–3 days before therapy; start ACE inhibitor at a low dose and observe the patient for several hours after the first dose.
Hypersensitivity reactions are more likely in patients with a history of asthma or allergies.
May be beneficial in hypertensive patients with osteoporosis due to its calcium-retaining effect.
Use in Pregnancy and Lactation
Pregnancy: Use of thiazide or thiazide-like diuretics during pregnancy is not recommended for treating edema or hypertension. These agents can cause reduced plasma volume, increased blood viscosity, and decreased placental perfusion, leading to fetal and neonatal complications such as bone marrow suppression, thrombocytopenia, and jaundice. Use only when clearly indicated and benefits outweigh risks.
Lactation: Chlorthalidone is excreted in breast milk. If treatment is necessary, breastfeeding should be discontinued to avoid potential adverse effects in the infant.
Driving and Operating Machinery: Chlorthalidone may cause dizziness or fatigue, especially at the start of therapy or with dose increases. Caution patients against driving or operating heavy machinery until they know how the drug affects them.
Overdose Management
Missed dose: If a dose is missed, take it as soon as remembered unless it is near the next scheduled dose. Do not double the dose.
Overdose symptoms:
- Dizziness, nausea, drowsiness
- Hypovolemia and hypotension
- Electrolyte disturbances (particularly hypokalemia), possibly leading to arrhythmias or muscle spasms
Treatment:
- No specific antidote available
- Perform gastric lavage, induce emesis, or administer activated charcoal to reduce absorption
- Monitor vital signs, serum electrolytes, and fluid balance
- IV fluid and electrolyte replacement as needed
- Symptomatic and supportive care is essential
Medical Disclaimer for Chlorthalidone Article:
The information provided in this article about Chlorthalidone is for educational and informational purposes only. It is not intended to replace professional medical advice, diagnosis, or treatment. Always seek the advice of your physician, pharmacist, or other qualified healthcare provider with any questions you may have regarding a medical condition or medication. Never disregard professional medical advice or delay seeking it because of something you have read on GoodMedToday.
If you experience any side effects, allergic reactions, or unusual symptoms while taking Chlorthalidone, contact your healthcare provider immediately.
