Hemophilia: A Dangerous Inherited Bleeding Disorder
Hemophilia is one of the most well-known inherited bleeding disorders, caused by a deficiency or absence of clotting factors VIII or IX. The severity of bleeding episodes depends on how much of these factors are missing. When bleeding occurs into muscles or joints, it may not appear immediately but can worsen within several hours after an injury.
Diagnosis is based on specific blood tests such as activated partial thromboplastin time (aPTT), prothrombin time (PT), and platelet count. The mainstay of treatment involves replacement therapy to restore the missing clotting factor, along with antifibrinolytic medications in certain situations to prevent recurrent bleeding.
Understanding Hemophilia
What Is Hemophilia?
Hemophilia is a genetic disorder that impairs the body’s ability to form blood clots, leading to prolonged bleeding after injury, surgery, or even spontaneously. Depending on the degree of factor deficiency, the condition can range from mild to life-threatening. In severe cases, uncontrolled bleeding may cause joint damage, anemia, and can even be fatal without prompt medical care.
There are several types of hemophilia, classified by the specific clotting factor that is deficient:
Hemophilia A (Factor VIII deficiency): The most common form of hemophilia, caused by a lack of clotting factor VIII. Patients often experience severe bleeding episodes into major joints such as the knees, elbows, or hips.
Hemophilia B (Factor IX deficiency, or Christmas disease): Caused by insufficient factor IX. Symptoms are similar to Hemophilia A and can vary in severity from mild to severe.
Hemophilia C (Factor XI deficiency): A rarer and generally milder form that can cause abnormal bleeding, particularly after surgery or trauma.
Signs and Symptoms of Hemophilia
People with hemophilia are prone to spontaneous or prolonged bleeding into tissues and joints (known as hemarthrosis), as well as bleeding within muscles or the retroperitoneal space. Depending on the level of clotting factor present in the blood, bleeding can occur immediately after trauma or be delayed for several hours.
By Severity:
Mild Hemophilia (Factor activity 5–25% of normal): Bleeding typically occurs only after significant trauma, surgery, or dental extraction.
Moderate Hemophilia (Factor activity 1–5% of normal): Bleeding may occur after minor injuries.
Severe Hemophilia (Factor VIII or IX activity <1% of normal): Spontaneous and frequent bleeding episodes, often beginning soon after birth.
Common Symptoms:
Joint bleeding (hemarthrosis): The most frequent symptom, usually affecting knees, ankles, or elbows, leading to swelling, pain, and limited motion. Repeated bleeding may cause chronic joint damage.
Muscle bleeding: May cause pain, swelling, and stiffness, sometimes compressing nearby nerves.
Internal bleeding: Such as bleeding in the gastrointestinal tract (esophagus, stomach, intestines) or urinary tract (hematuria).
Mucosal bleeding: Including frequent nosebleeds or bleeding from gums.
Heavy or prolonged menstrual bleeding in women with hemophilia or carriers of the gene.
When to See a Doctor
Seek medical attention immediately if you experience unusual bruising, prolonged bleeding, joint swelling, or unexplained pain. Early diagnosis and prompt treatment can prevent complications such as chronic joint damage or life-threatening hemorrhage.
With appropriate care—including prophylactic factor replacement and careful management of injuries—most individuals with hemophilia can lead active, healthy lives.
Causes and Risk Factors of Hemophilia
What Causes Hemophilia?
Hemophilia is a genetic bleeding disorder caused by mutations, deletions, or inversions in the genes responsible for producing clotting factors — primarily Factor VIII (in Hemophilia A) or Factor IX (in Hemophilia B).
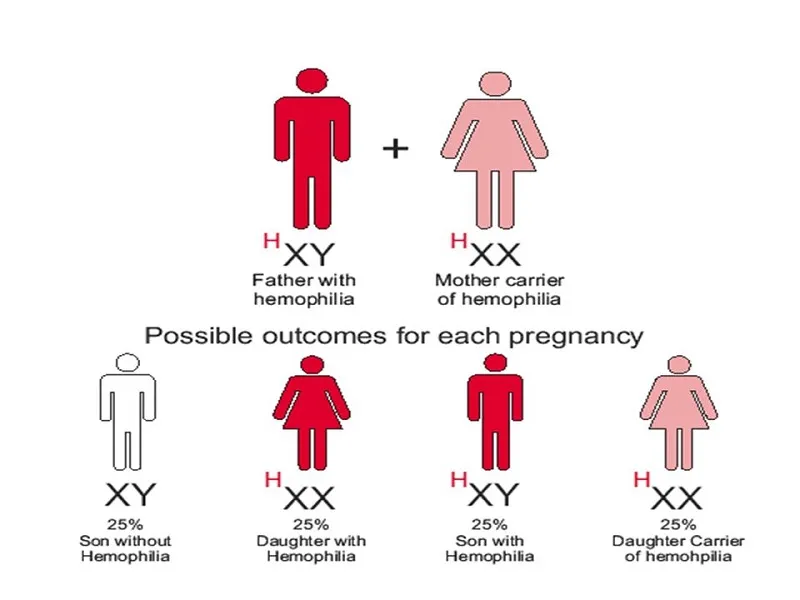
Because these genes are located on the X chromosome, hemophilia is classified as an X-linked recessive disorder, meaning it almost exclusively affects males, while females are typically carriers.
A man has only one X chromosome (inherited from his mother). If that X chromosome carries the defective gene, he will develop hemophilia. Women, on the other hand, have two X chromosomes — so even if one carries the mutation, the other usually compensates. However, they can pass the gene to their children.
- Daughters of men with hemophilia will always inherit the defective gene and become carriers.
- Sons of men with hemophilia will not inherit the disease, since they receive the father’s Y chromosome.
- Each son of a female carrier has a 50% chance of having hemophilia, and each daughter has a 50% chance of being a carrier.
While Hemophilia A and Hemophilia B follow this X-linked pattern, Hemophilia C (Factor XI deficiency) is inherited differently — it is autosomal, not linked to sex chromosomes, meaning both males and females can be equally affected. This type is more common in certain ethnic groups, such as individuals of Ashkenazi Jewish descent.
Who Is at Risk of Hemophilia?
Hemophilia primarily affects males due to its X-linked inheritance pattern. However, females can also develop symptoms in rare cases — especially if they inherit defective genes from both parents or experience X-chromosome inactivation that limits production of normal clotting factors.
Risk Factors Include:
- Family history of hemophilia:
Having a parent or close relative with the disorder significantly increases the risk. In fact, about two-thirds of all hemophilia cases are inherited. - Male gender:
Men are far more likely to have hemophilia, while women are typically carriers.
Interestingly, about one-third of new cases result from spontaneous genetic mutations, meaning there is no prior family history of the disorder. This highlights the role of de novo mutations that can arise unexpectedly during embryonic development.
Genetic Counseling and Prevention
Because hemophilia is hereditary, genetic counseling plays an important role for families with a known history of the disease. Carrier testing and prenatal diagnosis (via amniocentesis or chorionic villus sampling) can help identify whether the baby has inherited the gene mutation.
With modern reproductive technologies, such as preimplantation genetic testing (PGT) during IVF, at-risk families can plan pregnancies while minimizing transmission of the condition.
Diagnosis and Treatment of Hemophilia
Diagnostic Methods
When a patient presents with unexplained or recurrent bleeding and prolonged activated partial thromboplastin time (aPTT), hemophilia should be considered as a potential cause. Diagnosis typically involves a combination of screening and specific coagulation tests to confirm the deficiency and classify the type and severity.
1. Initial laboratory tests include:
- Platelet count
- Prothrombin time (PT)
- Activated partial thromboplastin time (aPTT)
- Assays for Factor VIII and Factor IX activity
In hemophilia, aPTT is prolonged, while PT and platelet count remain normal.
Further testing may include evaluation of von Willebrand factor (vWF) activity and antigen levels, since Factor VIII levels may also be reduced in von Willebrand disease (VWD).
2. Factor Assays:
Factor VIII assay helps diagnose and grade Hemophilia A.
Factor IX assay is used to confirm Hemophilia B.
For borderline or mild Hemophilia A, additional vWF studies (activity, antigen, and multimer analysis) help distinguish between Hemophilia A and VWD — especially when both males and females in the family have a history of bleeding.
3. Genetic testing and prenatal diagnosis:
For families with a known mutation, DNA analysis using polymerase chain reaction (PCR) techniques can identify the specific gene defect.
Prenatal diagnosis may be done via:
- Chorionic villus sampling (around the 12th week of pregnancy), or
- Amniocentesis (around the 16th week)
to detect mutations in the Factor VIII or IX genes.
Screening for Inhibitors
After repeated exposure to replacement therapy, some patients develop inhibitory alloantibodies that neutralize the infused clotting factors.
Approximately 30% of patients with severe Hemophilia A and 3% of those with Hemophilia B develop these inhibitors.
Therefore, inhibitor screening is essential before and during factor replacement therapy to optimize treatment outcomes.

Treatment Options for Hemophilia
Hemophilia is a lifelong genetic disorder, and while there is no definitive cure, treatment aims to control bleeding, prevent complications, and improve quality of life. The mainstay of therapy is replacement of the missing clotting factor, supplemented by modern biologic and gene-based approaches.
1. Replacement Therapy
Hemophilia A (Factor VIII deficiency)
Treatment involves intravenous infusion of plasma-derived or recombinant Factor VIII concentrates.
The required dose depends on the severity and site of bleeding:
| Clinical Situation | Target Factor VIII Level | Treatment Duration |
| Minor procedures (e.g., tooth extraction), early joint bleed | 30% of normal | 1–2 days |
| Moderate joint or muscle bleeding | 50% of normal | 3–5 days |
| Major surgery or life-threatening bleeding (intracranial, cardiac, etc.) | 100% of normal | Maintain ≥50% for 7–10 days |
Each 1 unit/kg of Factor VIII raises plasma levels by approximately 2%.
2. Monoclonal Antibody Therapy
Emicizumab is a bispecific monoclonal antibody that mimics the function of Factor VIII by bridging Factor IXa and Factor X, enabling clot formation without needing Factor VIII.
It can be administered subcutaneously and is highly effective for both prophylaxis and patients with inhibitors.
3. Desmopressin (DDAVP)
For mild to moderate Hemophilia A, Desmopressin (DDAVP) can temporarily increase endogenous Factor VIII and vWF release from endothelial cells.
It’s typically used:
- For minor surgeries or mucosal bleeding
- In patients with Factor VIII levels ≥5% of normal
However, it is not effective in Hemophilia B.
4. Hemophilia B (Factor IX deficiency)
Treatment uses Factor IX concentrates, either plasma-derived or recombinant, administered every 24 hours.
Because Factor IX has a larger volume of distribution, higher doses are needed compared to Factor VIII to achieve the same plasma level.
5. Antifibrinolytic Therapy
Adjunctive antifibrinolytics, such as:
- Tranexamic acid: 1.0–1.5 g orally 3–4 times daily for 5–7 days
- Epsilon-aminocaproic acid: 2.5–4 g orally 4 times daily for 1 week
These agents are useful to prevent recurrent or delayed bleeding after dental extractions or mucosal injuries, especially in the mouth and throat region.
6. Extended Half-Life Factor Products
Recent advances have produced recombinant fusion proteins and PEGylated factors with prolonged activity:
- Factor VIII-Fc fusion protein
- Factor IX-Fc fusion protein
- PEGylated Factor VIII or IX
These allow less frequent infusions, improving convenience and adherence.
7. Emerging Therapies
Two promising novel agents are under clinical investigation:
- Fitusiran: a small interfering RNA (siRNA) that suppresses antithrombin production, enhancing clot formation.
- Concizumab: a monoclonal antibody that inhibits tissue factor pathway inhibitor (TFPI), increasing thrombin generation.
Both represent non–factor replacement therapies offering easier administration and improved quality of life.
8. Gene Therapy
Gene therapy has opened a new era in hemophilia management.
Using adeno-associated viral (AAV) vectors, researchers can deliver functional Factor VIII or IX genes into liver cells, enabling long-term endogenous production of the clotting factors.
Clinical trials have shown sustained normalization of factor levels in many patients — suggesting the potential for a one-time, durable treatment.
9. Blood Transfusion Support
In cases of massive bleeding or severe anemia, blood transfusion may be necessary to stabilize blood pressure and preserve organ function. This is typically supportive rather than curative and should accompany factor replacement.
Modern hemophilia management has shifted from reactive treatment to preventive and personalized care.
With recombinant factors, biologic agents, and gene therapy now transforming the therapeutic landscape, patients with hemophilia can lead near-normal lives when managed properly under specialized care.
Lifestyle and Prevention for People with Hemophilia
Healthy Habits to Manage Hemophilia Effectively
Living with hemophilia requires a proactive approach that combines medical adherence, mindful living, and preventive care. While the condition cannot be cured, maintaining proper lifestyle habits can significantly reduce the frequency and severity of bleeding episodes, improve mobility, and enhance quality of life.
Daily Living Recommendations
Follow your doctor’s treatment plan carefully. Always take prescribed medication or factor replacement as directed. Never adjust your dosage or stop treatment without medical advice.
Maintain a positive and balanced lifestyle. Stress and fatigue can indirectly affect your overall health. Incorporate light exercise, adequate sleep, and stress-relieving activities such as yoga, meditation, or gentle stretching.
Stay alert to unusual symptoms. If you experience unexplained bruising, joint pain, or bleeding that does not stop easily, contact your doctor or go to a medical facility immediately.
Schedule regular medical checkups. Routine follow-up appointments allow healthcare providers to monitor your condition, adjust treatment plans, and prevent long-term complications such as joint damage.
Avoid injuries and sharp objects. Use protective gear when exercising and make your living space safe from hazards that may cause cuts or falls. In the event of serious bleeding that cannot be controlled, seek emergency care—do not attempt to self-treat with unproven remedies.
Nutritional Guidelines for Hemophilia
A balanced diet is crucial for maintaining overall health, supporting blood formation, and protecting joint function in people with hemophilia.
Recommended Foods
Soft, non-crunchy foods to prevent oral injuries (avoid hard bones, tough meat, or crispy snacks).
Iron-rich and calcium-rich foods such as leafy greens, pumpkin, potatoes, legumes, tofu, whole grains, lean red meat, and liver.
Low-fat dairy products like skim milk and cheese to strengthen bones and teeth.
Vitamin C–rich foods (oranges, tomatoes, bell peppers, strawberries, melons) help the body absorb iron more efficiently and support wound healing.
Foods to Limit or Avoid
Sugary beverages and processed foods (soda, energy drinks, sweetened tea).
High-fat or trans-fat foods including fried items, butter, pastries, pizza, and cookies.
Full-fat dairy products and animal fats, which may increase inflammation and weight gain—both harmful to joint health.
Maintaining a healthy body weight helps reduce stress on the joints, minimizing the risk of joint bleeding, which is one of the most common complications in hemophilia.
Preventive Measures for Hemophilia
Although hemophilia is a genetic disorder that cannot be fully prevented, several proactive measures can help reduce complications and protect patients from avoidable risks.
1. Genetic Testing and Counseling: If there is a family history of hemophilia, individuals—especially women who may be carriers—should undergo genetic screening and counseling before planning pregnancy. This helps assess the likelihood of passing the condition to children and prepares families for early intervention if needed.
2. Avoid Certain Medications: Patients should avoid aspirin and nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen or naproxen, as these can inhibit platelet function and increase the risk of bleeding. Always consult a doctor before taking any new medication, including herbal or over-the-counter products.
3. Oral and Dental Care: Maintain excellent oral hygiene to prevent gum disease and tooth decay, reducing the need for invasive dental procedures. Use a soft toothbrush and visit a dentist familiar with bleeding disorders for preventive care.
4. Injection and Medication Safety: Whenever possible, avoid intramuscular (IM) injections, which can lead to deep muscle bleeding and bruising. Opt for oral or intravenous (IV) administration when medication is necessary.
5. Vaccinations: People with hemophilia should receive vaccines for hepatitis A and B.
Although plasma-derived clotting factors are screened and treated for viruses, residual risk of viral transmission (such as parvovirus B19 or hepatitis) still exists. Immunization adds an extra layer of protection.
6. Physical Activity: Engage in low-impact exercises such as swimming, walking, cycling, or stretching to strengthen muscles and joints while minimizing trauma risk. Avoid contact sports like football, hockey, or boxing that can cause internal bleeding or fractures.
7. Emergency Preparedness: Patients and families should learn how to recognize early signs of bleeding and administer factor replacement promptly when necessary. Wearing a medical alert bracelet can help ensure appropriate treatment in emergencies.
By following a tailored plan that includes safe habits, nutritious eating, and regular monitoring, individuals with hemophilia can lead active, fulfilling, and largely normal lives.
